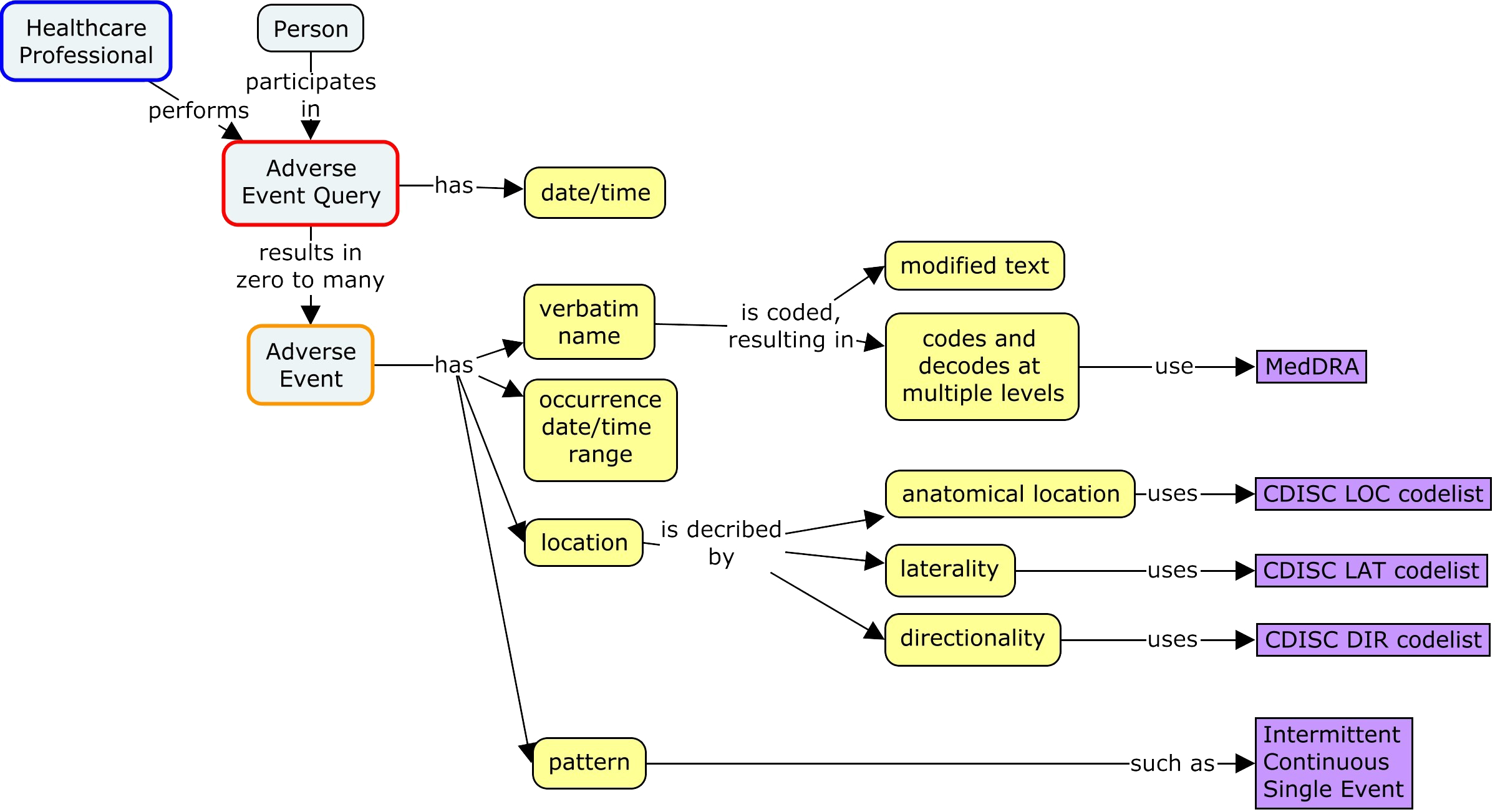
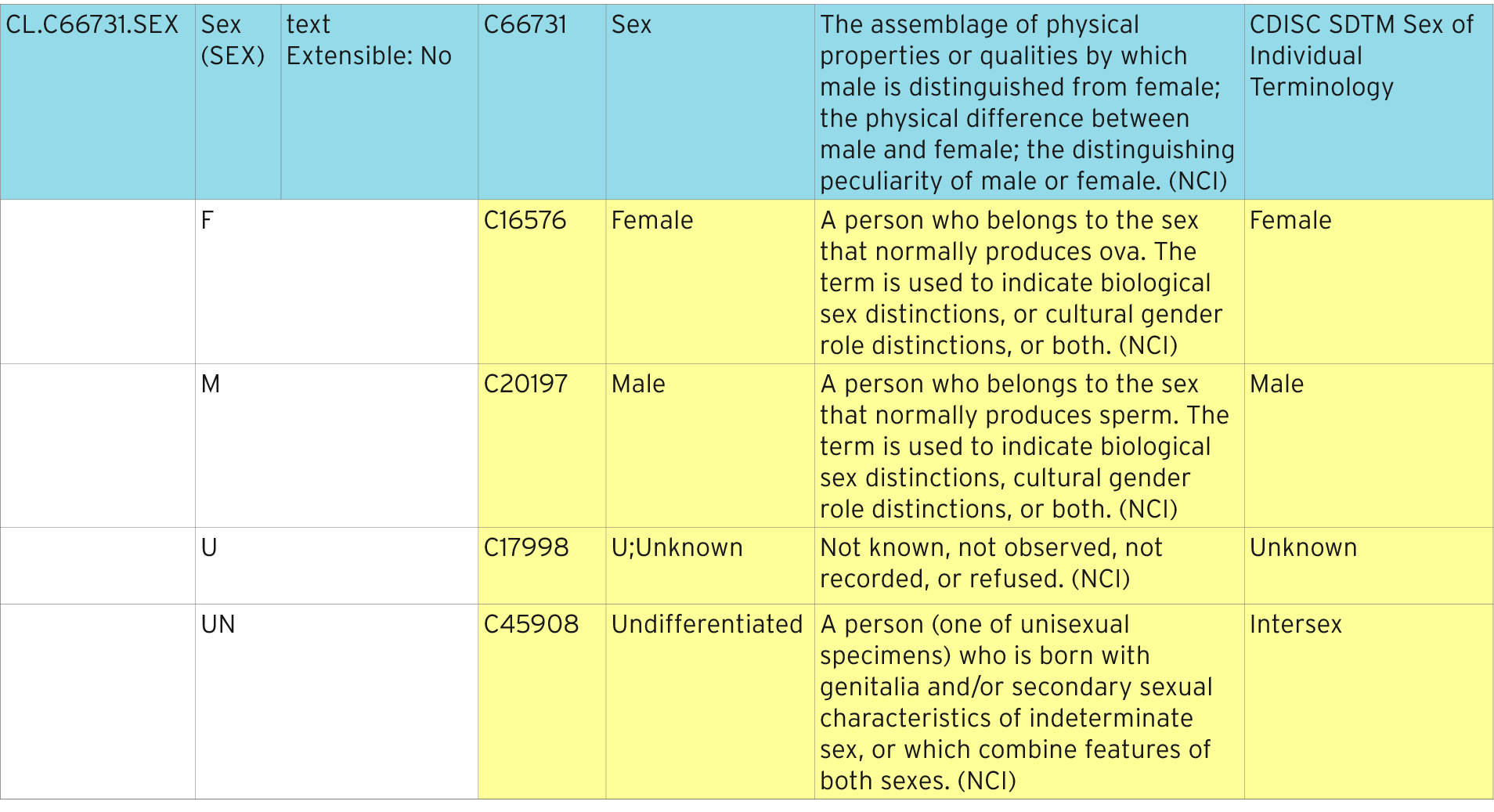
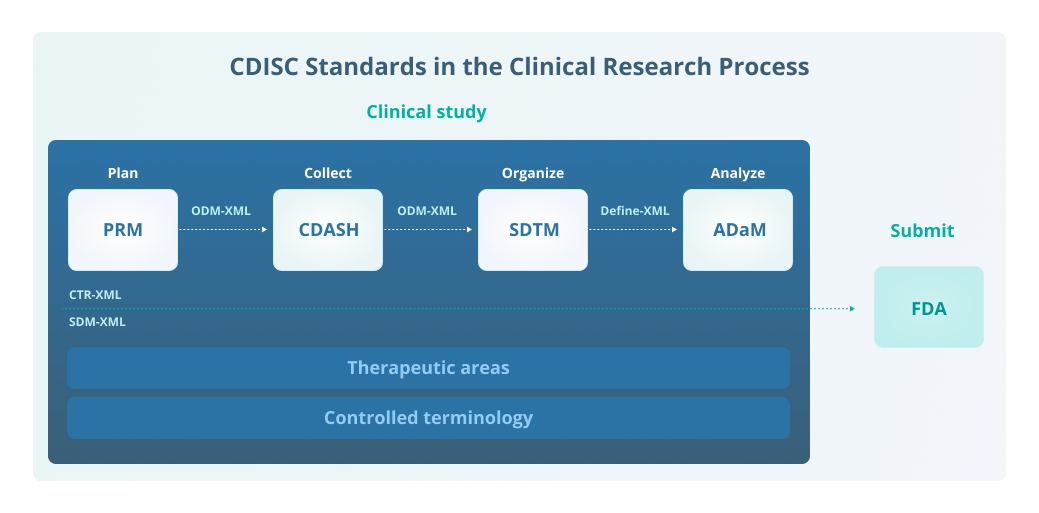
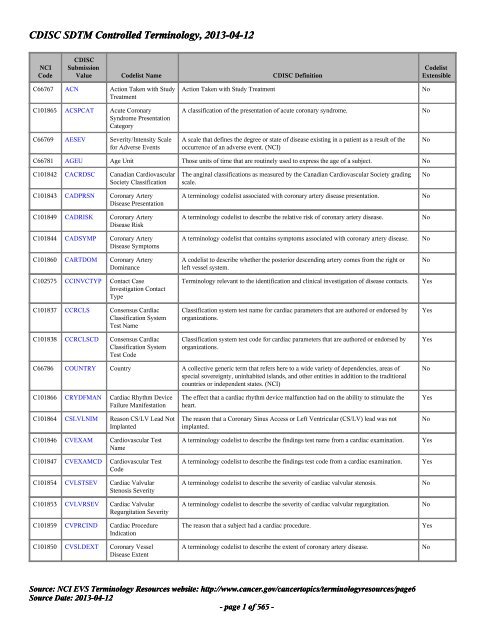
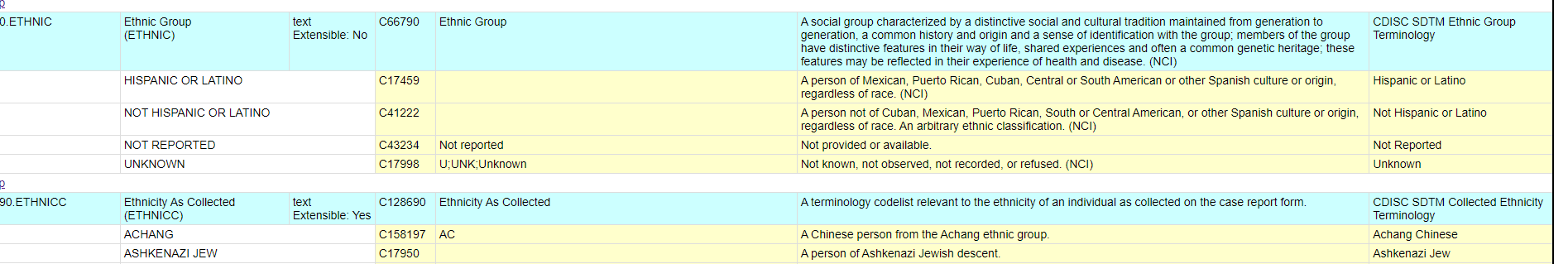
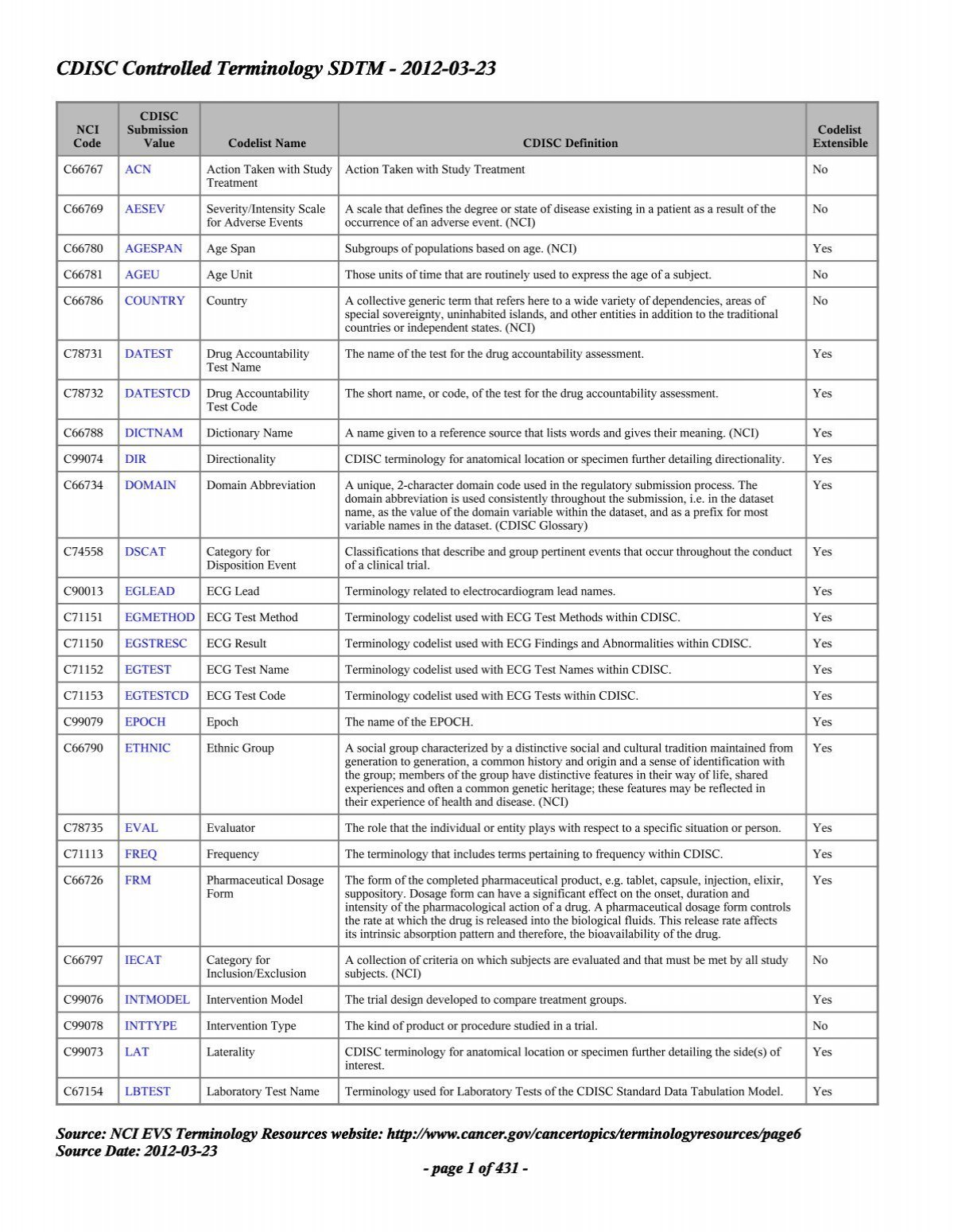
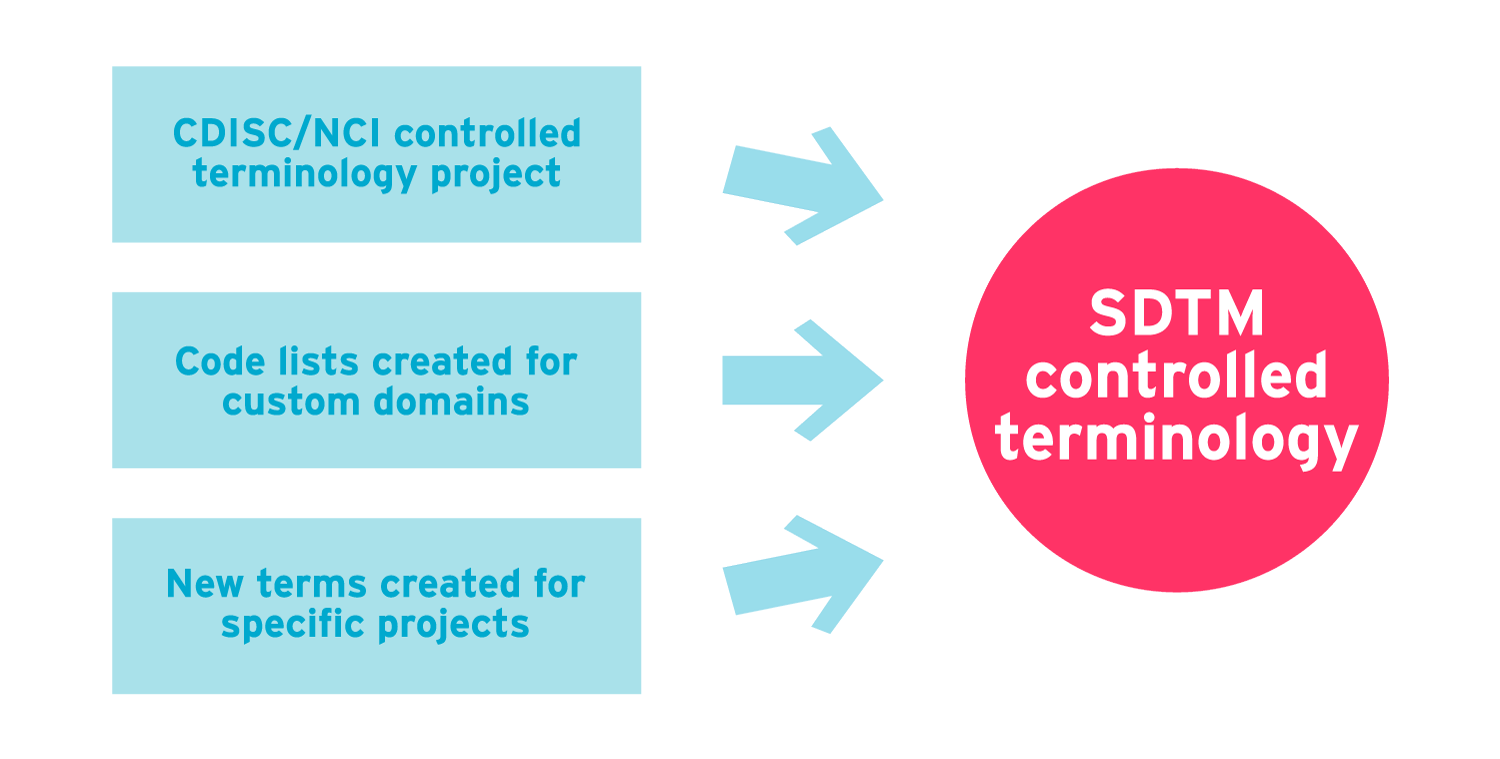
Controlled Terminology for Clinical Research: A Collaboration Between CDISC and NCI Enterprise Vocabulary Services
173-2012: Automatic Consistency Checking of Controlled Terminology and Value Level Metadata between ADaM Datasets and Define.xml

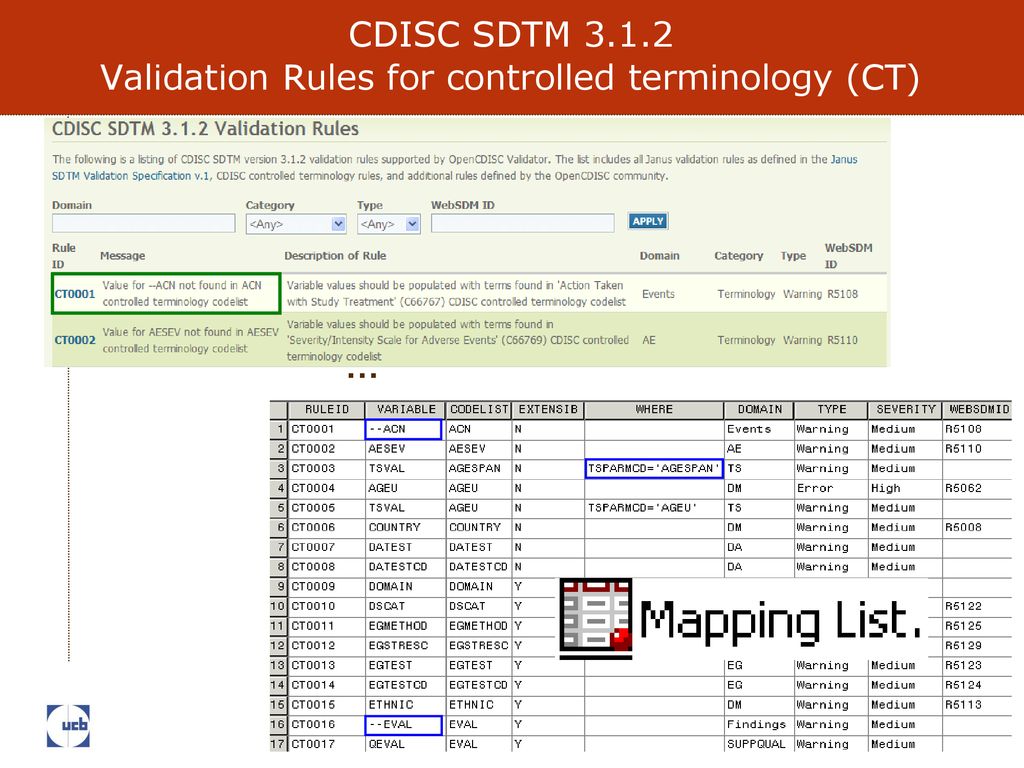
Figure 1 from From ACE to ZINC Examples on the use of SDTM Controlled Terminology for lab tests | Semantic Scholar